Our Timeline
At MED-EL, our pioneering spirit and passion for discovery has been going strong for more than 40 years. We’re proud to be the innovation leader, with technological breakthroughs and one-of-a-kind developments coming year after year.

The Beginning
Ingeborg and Erwin Hochmair begin developing cochlear implants at the Technical University of Vienna.
1977

First Modern Cochlear Implant
The world's first microelectronic multi-channel cochlear implant in Vienna. It had 8 channels, a stimulation rate of 10,000 pulses per second per channel, 8 independent current sources, and a flexible electrode for 22-25mm insertion into the cochlea.

Prof. Kurt Burian (1924-1996)
Implanted the first multichannel microelectronic cochlear implant at the ENT University Clinic in Vienna on December 16th 1977. He inserted the flexible electrode through the round window into the scala tympani.
1978


6- and 4-channel implant variants
The 4-channel implant was used for simultaneous stimulation.
1979

Early Success
C.K., received a 4-channel implant in August 1979 and a small portable processor a few weeks later. She spent many hours in the test lab at the Technical University of Vienna where a number of speech coding strategies and stimulation field configurations were tested for fundamental research on nerve stimulation.

Fine Tuning

Sound Optimisation
1980

Understanding Speech
1982

Further Development
This Cochlear Implant System consists of a signal-transparent 4-channel implant driving a long intra-cochlear 4-channel flexible electrode and a small single-channel speech processor. The system implements a broadband analogue speech coding strategy to be used on the best-suited channel.
1985

Getting Together
1986
Moving to Innsbruck
1989


Comfort CI System
Introduction of the Comfort CI System: The new implant electronics and coils are now encased in one (2-channel system) or two (4-channel system) hermetically sealed ceramic housings. The new speech processor features an ear-level microphone.
Over the years around 700 adults and children received this system, including the early variant with pacemaker epoxy encapsulation.
The majority of these recipients are now using contemporary implants and enjoy further improved speech understanding.
1990
First Employees
1991

First BTE Audio Processor
MED-EL launches the world's first BTE (behind-the-ear) speech processor. It provides the same speech understanding as the body-worn speech processor.

First CAP Implant
M.H. is the first to receive an implant that combines analogue and pulsatile stimulation.
1994


COMBI 40 System
The implant system COMBI 40 is launched. It is the world's first multichannel high-rate CI, an 8-channel system designed to faithfully implement Blake Wilson's CIS speech coding strategy with high stimulation rate.
It comprises a 31 mm long flexible electrode for coverage of the entire length of the cochlea.
1995

Excellent Results
Multicentre study results with the implant COMBI 40 in post-lingually deaf adults demonstrate for the first time that more than half of the patient population reached a monosyllabic word understanding higher than 50% after only 6 months. This means that these patients can talk to someone unknown to them about an unknown topic over the telephone.

The CIS PRO+ Processor

CIS LINK
Out of humanitarian reasons an addition to the CIS PRO+, the CIS LINK system, is developed to provide the CIS strategy to Ineraid recipients who had received percutaneous plug CI systems by another company which discontinued further development.
1996

Introduction of the COMBI 40+
The thinnest cochlear implant available. It provides up to 50,000 pulses per second and is a miniaturised multichannel implant (4 mm thin).
First Bilateral Implantation
1997


More Surgical Options for Rare Cases
The MED-EL Auditory Brainstem Implant is introduced for the first time. The ABI allows people without a functioning auditory nerve and NF2 to access sound.
1998

First Bilateral Child
1999

TEMPO+ BTE Audio Processor
Launch of the TEMPO+ Behind-the-Ear (BTE) Speech Processor.
It consumes 90% less power than the body-worn processor.
A multicentre study demonstrates speech perception results superior to the body-worn processor.
The TEMPO+ is the first processor to have a modular design allowing different wearing options.
Modular BTE processor design makes the body-worn processor obsolete.
2003

VIBRANT SOUNDBRIDGE
Acquisition of VIBRANT SOUNDBRIDGE, the first active middle ear implant system for mild-to-severe sensorineural hearing loss.
Geoffrey Ball, the inventor of the VIBRANT SOUNDBRIDGE, moves to Innsbruck and becomes CTO of VIBRANT MED-EL.
2004

PULSAR Cochlear Implant
Launch of the PULSAR Cochlear Implant, providing future-ready electronics in an optimised ceramic housing.
2005


First EAS System
Introduction of the DUET Speech Processor, the world's first processor to integrate cochlear implant speech processing and acoustic stimulation in one compact device.
The EAS System is the world's first hearing implant system for combined acoustic and electric stimulation. Electric acoustic stimulation (EAS) is a concept that was started by Prof. Christoph von Ilberg with his first EAS patient successfully implanted in 2000.
2006


Driving Innovation
The next generation of cochlear implant systems is launched. The OPUS Audio Processor has a sleek, switch-free design; the titanium SONATA Cochlear Implant offers exceptional reliability; FLEX electrode arrays help protect natural hearing structures; and FineHearing technology provides more natural sound perception.
2007

DaCapo Rechargeable Battery System
2008
Expanding Treatment
VIBRANT SOUNDBRIDGE receives the CE mark for an expanded range of candidacy, including conductive and mixed hearing loss.
2009

Second Generation EAS
The DUET 2 EAS Audio Processor is launched. This second generation EAS audio processor combines acoustic amplification and cochlear implant stimulation, bringing sound to people with high-frequency hearing loss.

Amadé for SOUNDBRIDGE
2010

MAESTRO CI System
MED-EL launches the new MAESTRO Cochlear Implant System. MAESTRO features the OPUS 2 Audio Processor and the CONCERTO Cochlear Implant.

CONCERTO ABI
The CONCERTO Auditory Brainstem Implant (ABI) is introduced. This enables people without a functioning auditory nerve to perceive sound using the latest hearing technology.
2011

FLEX Electrode Arrays
The new FLEX 28 Electrode Array is introduced. It’s made for better structure and hearing preservation, to provide superior hearing performance.

Vibroplasty Couplers for VIBRANT SOUNDBRIDGE
New couplers for the VIBRANT SOUNDBRIDGE give surgeons even more flexibility with the middle ear implant.
Mini Battery Pack
2012

First Active Bone Conduction Implant
Launch of BONEBRIDGE, the world’s first active bone conduction implant system that leaves the skin intact.

Smallest & Lightest Audio Processor
2013

RONDO Single-Unit Processor
Just put it on and go: RONDO is the first single-unit cochlear implant audio processor. The all-in-one design combines the control unit, cable, and coil into one compact single unit.

FORM Electrode Arrays

First for SSD
MED-EL CI systems are the first CIs approved for treating single-sided deafness in both adults and children.

AUDIOVERSUM Opens
MED-EL opens its own ScienceCenter, AUDIOVERSUM, in Innsbruck, Austria. With its interactive, multimedia exhibitions, visitors can discover the wonders of hearing, and take part in a range of special events and workshops.
2014

SYNCHRONY CI System
MED-EL continues to drive innovation with the SYNCHRONY Cochlear Implant System.
The SYNCHRONY Cochlear Implant offers MRIs at 3.0 Tesla. That’s because the magnets in our SYNCHRONY implant can freely rotate and self-align inside of its housing so it’s not adversely affected by the magnetic field of an MRI scan.
The splash-proof SONNET Audio Processor with Automatic Sound Management 2.0 enables recipients to hear effortlessly in any environment.

SOUNDBRIDGE (VORP 503) and SAMBA
VIBRANT SOUNDBRIDGE (VORP 503) is upgraded for offering improved ease-of-use. Along with the new Vibroplasty Couplers, surgeons have increased flexibility when implanting SOUNDBRIDGE. The new implant is also MRI Conditional at 1.5 Tesla.
SAMBA is released as the latest audio processor for SOUNDBRIDGE and BONEBRIDGE.
2015
New Child-Friendly Features

Lightest Audio Processor
2017

Lightest and Smallest Wearing Option
ActiveWear, the lightest and smallest wearing option available, is released for SONNET, along with BabyWear. Both make SONNET more comfortable and secure for babies, children, and active adults.

Wireless Connectivity for SONNET
The Roger 21™ from Phonak is launched for SONNET. It allows SONNET users to use Roger wireless microphones and connect to their phone, TV, and other devices.


ADHEAR Bone Conduction System
The novel, non-surgical ADHEAR hearing system revolutionises the concept of bone conduction for people with conductive hearing loss and single-sided deafness.

RONDO 2 Audio Processor
RONDO 2 is launched. RONDO 2 is the first cochlear implant audio processor which can be charged wirelessly, making it incredibly simple to use.

Expanded Indication for ABI
2018

OTOPLAN – MED-EL Supports Leading Surgical Technology
OTOPLAN is launched. This revolutionary software manufactured by CAScination allows surgeons to quickly generate patient-specific 3D reconstruction from CT images. They can easily visualize each patient’s unique anatomy before entering the operating room helping to create the best outcomes during the surgery itself.
2019

New and Improved SYNCHRONY System

BONEBRIDGE BCI 602 Implant
2020

SAMBA 2 Audio Processor

RONDO 3 Audio Processor
RONDO 3 is launched. With wireless streaming, wireless charging and superior hearing performance, RONDO 3 is incredibly simple for cochlear implant recipients to use. RONDO 3 can even be easily adjusted for optimal hearing with any brand of hearing aid on the recipient's other ear.

Direct Streaming With AudioStream
The launch of AudioStream makes streaming even simpler for SONNET series users. The AudioStream cover offers direct, wireless streaming from compatible phones. With it, MED-EL recipients can enjoy music and phone calls in high-quality, true stereo.

Passive Middle Ear Implants
A new product line is introduced: passive middle ear implants (PMEIs). This allows MED-EL to offer a complete portfolio of hearing implants and affirm its position as the world’s leading hearing implant company.
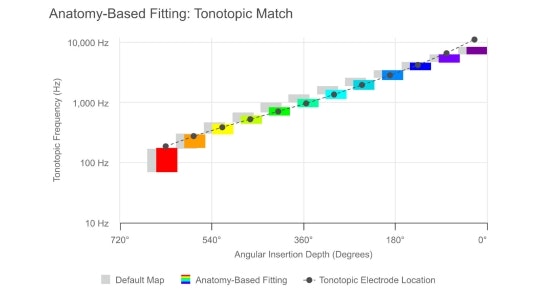
Anatomy-Based Fitting
Innovations in the MAESTRO fitting software and OTOPLAN surgical software enable more precise fitting. By matching tones to their natural places based on the unique anatomy of each ear, this helps MED-EL cochlear implant users achieve the closest to natural hearing.
2021
MRI Guarantee

30 Years of Innovation
The compendium "30 Years of Translational Research Behind MED-EL" was published, initiated, and coauthored by Ingeborg Hochmair. It reflects on decades of cochlear implant and ABI research, expresses appreciation for the key role of pioneers and their collaboration, and provides a glimpse into the future.
2022

Sound Sensation Music Festival
2024

Direct Streaming for RONDO 3
The AudioStream Adapter provides direct wireless streaming for RONDO 3. When plugged into RONDO 3, it streams music and speech in high-quality true stereo from tablets and smartphones. The compact AudioStream Adapter offers a convenient solution for streaming music, talking on the phone, and watching TV.

Remote Care With the HearCare MED-EL App
The HearCare MED-EL app is launched, making hearing care more convenient for RONDO 3, SONNET 3, SONNET 3 EAS, SONNET 2, and SONNET 2 EAS users. With innovative features, such as remote fitting without an appointment and configuration backups in the cloud, it is even easier for cochlear implant recipients to hear their best.

SONNET 3 Audio Processor
SONNET 3 is launched. SONNET 3 is our smallest and lightest SONNET, equipped with integrated direct streaming, a flexible silicone earhook, and an innovative touch key.
2025

New Passive Middle Ear Implants
Our selection of passive middle ear implants for tympanoplasty and stapes surgery is expanded from 11 to 16 with new options to suit each patient’s individual ear anatomy.
2026

New Rechargeable Batteries for SONNET 3
With the Micro Pro and Standard Pro rechargeable batteries, SONNET 3 audio processor users can now experience more hearing time in a smaller and lighter package.

Looking Forward
Today, our dedicated research & development continues to drive discovery and innovation in the field of hearing and beyond. The future holds so much potential, and we're excited to be at the forefront of incredible breakthroughs and leaps in technology.