Bone Conduction Implant
BONEBRIDGE gives you great hearing, but that’s not all. It is the most advanced active bone conduction implant and is fully placed under the skin. In fact, you won’t even realize it’s there! Find out how BONEBRIDGE uses bone conduction to revolutionize your hearing.


Comfortable and Effective
BONEBRIDGE is an implant designed with your lifestyle in mind. Because the implant is fully under your skin, BONEBRIDGE doesn’t leave you with open wounds or skincare routines to worry about. The externally-worn SAMBA 2 Audio Processor sits comfortably on your head and its streamlined design makes it easy to hide under your hair. You can even connect it to your phone via SAMBA 2 Go.
But most importantly, BONEBRIDGE offers you great sound quality. MED-EL pioneered “under-the-skin” bone conduction implants when we introduced BONEBRIDGE over ten years ago. Thousands of happy users around the world show just how successful BONEBRIDGE is. And we’ve even combined this market experience with feedback from leading surgeons to create a second-generation BONEBRIDGE implant. For you, that means high-quality, comfortable hearing that you can rely on for years to come.
Natural sound quality
Placed fully under the skin
Small and stylish audio processor

Awarded 2019 to the BONEBRIDGE BCI 602 Bone Conduction Implant.

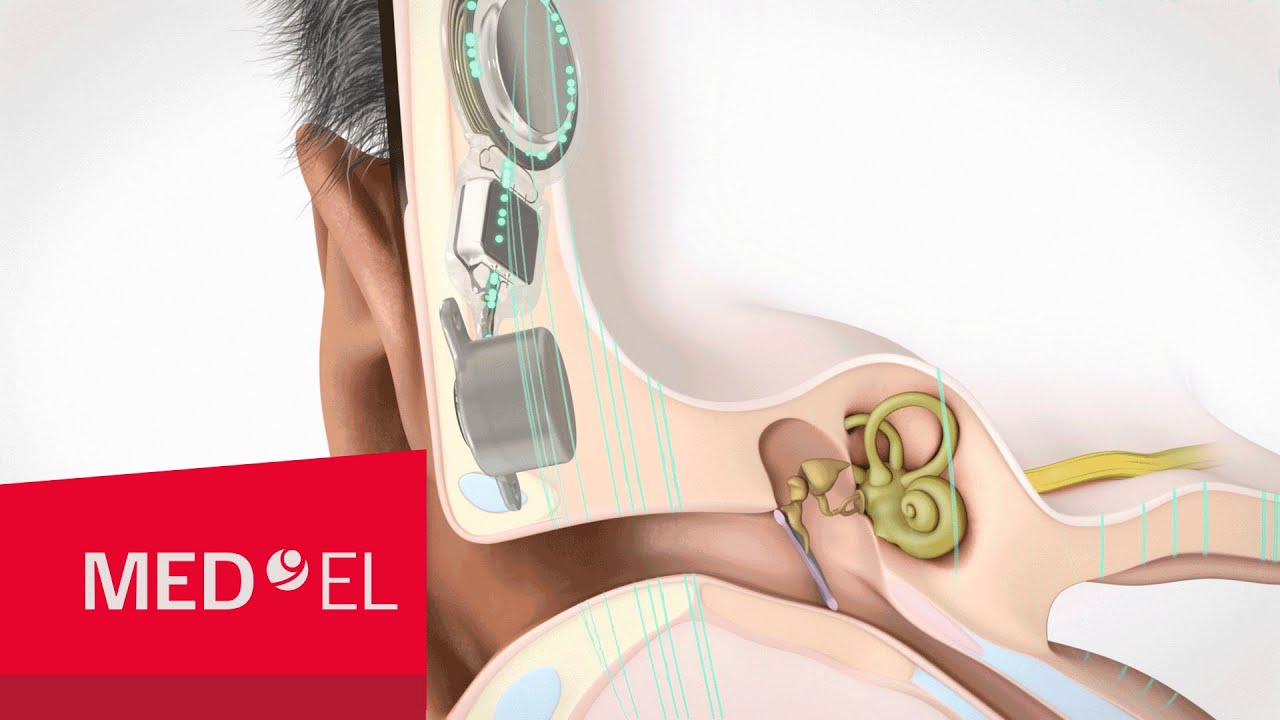
How Does BONEBRIDGE Work?
With normal hearing, sound vibrations are sent through the outer and middle parts of the ear and on to the inner ear. If you have conductive or mixed hearing loss, your outer or middle ear is damaged, so sounds can't reach your inner ear effectively.
But BONEBRIDGE can help. BONEBRIDGE picks up the sounds around you and sends them to your inner ear through the bones of your skull, bypassing your damaged outer and middle ears. If you have single-sided deafness, BONEBRIDGE can also be used to pick up sound vibrations on your bad side and send them through to the inner ear on your good side. The result? BONEBRIDGE restores the world of sound that is missing when you have hearing loss.

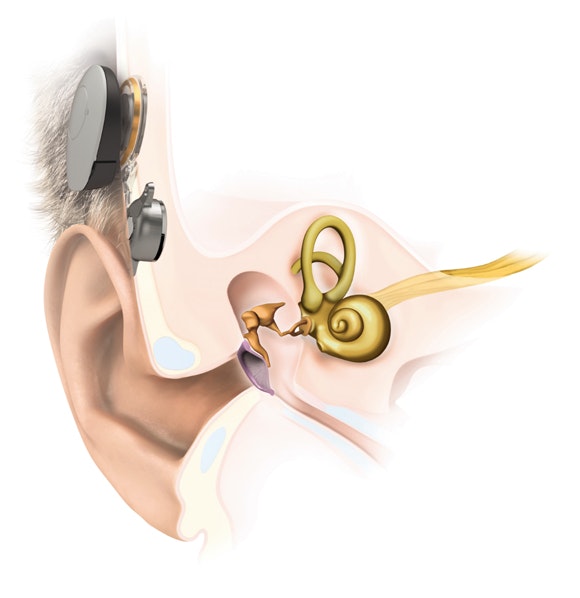
SAMBA 2
Audio Processor
- Small and light audio processor
- Stylish design covers
- Bluetooth® streaming via SAMBA 2 Go

BONEBRIDGE
BCI 602 Implant
- Natural sound quality
- Placed fully under the skin
- 1.5 Tesla MRI* without surgery^
SAMBA 2 Audio Processor
SAMBA 2 is the latest generation of audio processor for BONEBRIDGE. With a sleek, intuitive design and intelligent hearing technology. It's designed to make your hearing experience as simple as possible.
Comfortable to Wear
BONEBRIDGE is an active bone conduction implant that is fully implanted under the skin. For you, that means nothing sticking out through your skin, no open wounds, and therefore no daily skin care routines to worry about. You can simply put on your SAMBA 2 Audio Processor each morning and forget about it for the rest of the day.

A Stylish Solution
Make BONEBRIDGE invisible. The SAMBA 2 Audio Processor is worn on your head just behind your ear and is held comfortably in place by magnets. This makes it easy to hide the compact SAMBA 2 under your hair. Or you can choose to stand out from the crowd with SAMBA 2's colorful Design Covers.
Made for the Future
A hearing implant is a decision for a lifetime, so it is important to choose a system you can rely on. BONEBRIDGE’s robust design is built to keep you hearing for many years to come. The implant can undergo MRI scans at 1.5 Tesla* without discomfort, surgery, or hearing downtime,^ so you’re prepared for whatever the future might bring. What’s more, MED-EL creates audio processors that are compatible with previous implant models. So BONEBRIDGE recipients can easily upgrade to the newest audio processor and benefit from the latest in hearing technology.


Great Protection. Guaranteed.
Because of our long and positive experience with MRIs, we offer a life-long MRI guarantee.**
This life-long MRI guarantee is the first and only to be offered by any hearing implant company and covers all MED-EL BONEBRIDGE implants. So you can feel confident about any future MRI scans.
Experiences with BONEBRIDGE
BONEBRIDGE users tell us how their bone conduction implant has not only improved their hearing, but also their lives—at work, at home, and with friends and family.
“Today, I still can’t believe what a difference this small device can make!"
Daniela A., BONEBRIDGE user from Austria
"It felt so natural, like I had been wearing it for years."
The Next Steps
What can you expect next? Here’s an outline of the process of receiving a bone conduction implant.
1
Assessment
2
Implantation
To implant BONEBRIDGE, the surgical procedure is straightforward and usually lasts around 1-2 hours under general anesthesia. Recipients are usually back on their feet the next day. Your hospital stay may last one or more days, depending on your individual needs and what’s typical in your area.
3
Activation
As soon as the surgical area has healed, you’ll have your first fitting. This “activation day” is when your audiologist will turn on your audio processor for the first time and you’ll hear your first new sounds. The audio settings of your processor will be adjusted to fit your hearing preferences.
* The BONEBRIDGE bone conduction implant is MR conditional. Recipients with BONEBRIDGE may be safely MRI scanned at 1.5 Tesla following the conditions detailed in the instructions for use.
** The terms and conditions of the MRI Guarantee can be found here.
^ Unless required for diagnostic reasons.
Technical Data


BONEBRIDGE
Bone Conduction Implant (BCI 602)
Weight
Approx. 20 g
Features
- Transcutaneous technology
- The implant including the transducer is safely embedded under the skin
- The risk of skin infection is very low and no continuous skin care is needed
- Direct drive bone conduction together with state-of-the-art signal processing provides excellent audiological outcomes
- Cosmetically appealing solution
MRI Safety
MR Conditional at 1.5 Tesla
The Bluetooth® word mark and logos are registered trademarks owned by the Bluetooth SIG, Inc. and any use of such marks by MED-EL is under license.
Contact Us
Don’t wait any longer to take the next step. Our team of hearing professionals is ready to answer any questions you may have to help you decide what is right for you.
Get in Touch
Would you like more information on our hearing loss solutions? Just fill out our simple contact form and we’ll get back to you.